Chemistry - it's all about the particles! How are they arranged? What will they do next? HAVE THEY JOINED UP IN COMPOUNDS OR ARE THEY JUST TOO LAZY?
|
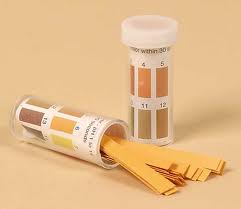
What do you remember about acids and alkalis from Year 7? It was a while ago, that's for sure, so maybe try this one out as a refresher.
|
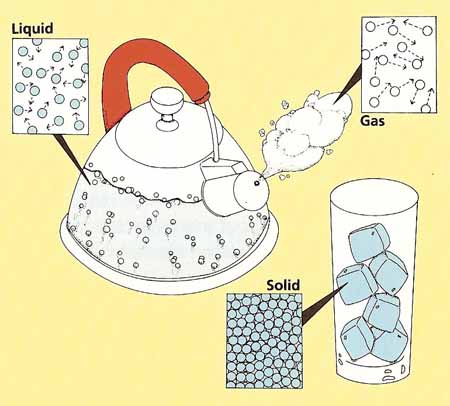
Good old BBC Bitesize. This is the main activity on how particles are arranged in the three main states of matter. Remember the better you know this section of the topic, the more the rest will make sense.
|
|
A favourite site for visual learners. This is a collection of great images for each element as well as the usual basic data about the element. Keep scrolling down through the images once you have selected your element and you never know what you might find.
|
Try out the little simulation on states of matter at home! We used this in class but sometimes your learning is better if you try things out for yourself. Do you learn like this?
|
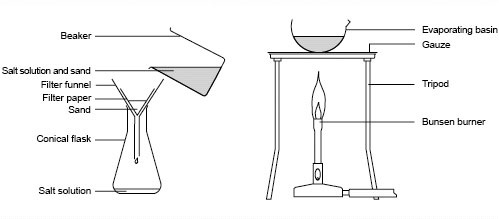
separating rock saltA solution of salt and water with sand is in the beaker. The sand is insoluble so can be separated from the salty water solution by filtering.
The salt is soluble in water so the salt solution is heated allowing the water to evaporate. This leaves salt crystals behind in the evaporating basin. |
|
|
qwcDescribe the processes that you would use to extract pure salt from rock salt. Include the apparatus you would use. [6 marks]
This is an example of the type of written answer you could be asked. QWC stands for Quality of Written Communication so it tests if you can write clearly using scientific terms and if your answer is organised logically. Take care with your spelling, punctuation and grammar too just as you would in an English essay! |
DistillationA easy way to separate a mixture of two liquids is to use distillation. This uses the fact that different substances will have different boiling points. So we can heat until the first substance A reaches its boiling point. This undergoes its physical change into its gaseous state and spreads through the condenser tube. Meanwhile, the second substance B will still be a liquid and remains in the flask. The Leibig condenser tube is cooled by passing cold water (cheap and ever available) through the outside part of the tube. Cooling the gas A inside the tube lowers its temperature below the boiling point so it will condense to its liquid state again. Drops of pure substance A are collected in a beaker.
|
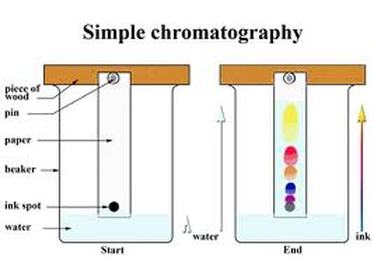
chromatographyChromatography is one of the most valuable techniques biochemists have for separating mixtures. It can be used to determine the ingredients that make up a particular flavor or scent, to analyze the components of pollutants, to find traces of drugs in urine, and to separate blood proteins in various species of animals (a technique that's used to determine evolutionary relationships) It is also used by forensic scientists.
(above text from http://www.exploratorium.edu/science_explorer/black_magic.html) Food scientists also use chromatography to determine if colours used are legal for use. Most food colours have E numbers and must be safe for consumption. It is more common now to use extracts from natural plants (like beetroot to give a red colour) than to use a completely artificial colour. Check the ingredients list on your sweeties to see what you can find out! |
|
Once you get the hang of the gridlocks game format, it helps you learn common formulae. This is always going to be useful in your studies of chemistry, no matter what level you get to.
|
A second version of the gridlocks game and even more important than learning useful formulae. Here you start to match up symbols and elements. As you go up the levels it gets harder.
|
This third gridlocks game is good for keeping you safe in the lab. Match up hazard symbols and practice logical thinking too.
|
Photo used under Creative Commons from ricardodiaz11